
Citation
S. Hu, Y.‐H. Tee, A. Kabla, R.Zaidel‐Bar, A. Bershadsky and P. Hersen
Cytoskeleton 72(5):235–245 (2015)
Abstract
Abstract
The ability to mechanically interact with the extracellular matrix is a fundamental feature of adherent eukaryotic cells. Cell-matrix adhesion in many cell types is mediated by protein complexes called focal adhesions (FAs). Recent progress in super-resolution microscopy revealed FAs possess an internal organization, yet such methods do not enable observation of the formation and dynamics of their internal structure in living cells. Here, we combine structured illumination microscopy (SIM) with total internal reflection fluorescence microscopy (TIRF) to show that the proteins inside FA patches are distributed along elongated subunits, typically 300±100 nm wide, separated by 400±100 nm and individually connected to actin cables. We further show that the formation and dynamics of these linear subunits are intimately linked to radial actin fiber formation and actomyosin contractility. We found FA growth to be the result of nucleation of new linear subunits and their coordinated elongation. Taken together, this study reveals that the basic units of mature focal adhesion are 300 nm-wide elongated, dynamic structures. We anticipate this ultrastructure to be relevant to investigation of the function of FAs and their behavior in response to mechanical stress. This article is protected by copyright. All rights reserved.
Figure sample
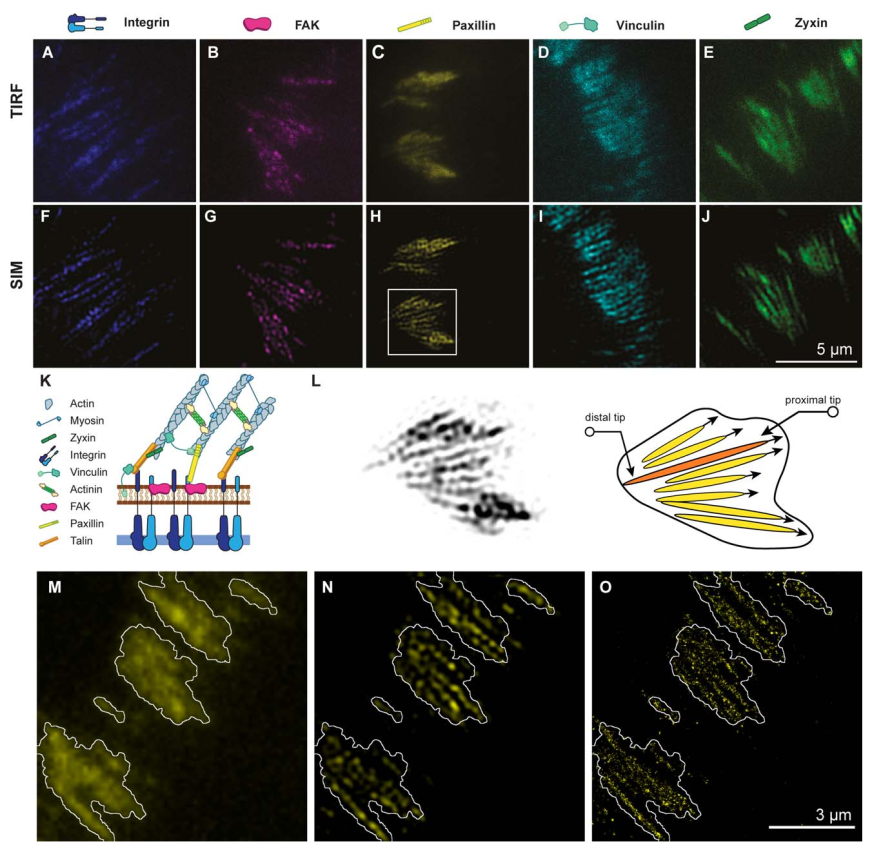
Focal adhesions are composed of arrays of linear subunits. A–J: Images of integrin b1-eGFP, FAK–GFP, paxillin–YFP, vincu- lin–mCherry, and zyxin–GFP obtained using total internal reflection microscopy (TIRF; A–E) and structured illumination micros- copy–total internal reflection microscopy (SIM–TIRF; F–J). Focal adhesion proteins were observed as large, relatively homogeneous patches using TIRF, whereas SIM–TIRF reveals them to be composed of an array of linear, parallel subunits. K: Illustration of the vertical organization of a focal adhesion. L: Magnified view of panel H (left) and sketch of the internal structure of this patch (right). The FA plaque (black contour) is composed of an array of linear subunits which extend toward the center of the cell. The proximal tip of the FA subunit is defined as the FA tip closer to cell center, and the distal tip is the FA tip closer to cell edge. M–O: REF52 cells expressing the photoswitchable protein paxillin–tdEos were fixed and imaged using three different microscopy techniques: TIRF (M), SIM–TIRF (N), and stochastic optical reconstruction microscopy (STORM, O), confirming that SIM–TIRF provides a high fidelity picture of the linear subunits observed using STORM. The white contour indicated the localization of the FA plaque as seen by TIRF (M).

