
Citation
N. Khalilgharibi, J. Fouchard, N. Asadipour, R. Barrientos, M. Duda, A. Bonfanti, A. Yonis, A. Harris, P. Mosaffa, Y. Fujita, A. Kabla, Y. Mao, B. Baum, J. Muñoz, M. Miodownik and G. Charras
Nature Physics 15:839–847 (2019)
Abstract
Abstract
Epithelial monolayers are one-cell-thick tissue sheets that line most of the body surfaces, separating internal and external environments. As part of their function, they must withstand extrinsic mechanical stresses applied at high strain rates. However, little is known about how monolayers respond to mechanical deformations. Here, by subjecting suspended epithelial monolayers to stretch, we find that they dissipate stresses on a minute timescale and that relaxation can be described by a power law with an exponential cut-off at timescales larger than about 10 s. This process involves an increase in monolayer length, pointing to active remodelling of cellular biopolymers at the molecular scale during relaxation. Strikingly, monolayers consisting of tens of thousands of cells relax stress with similar dynamics to single rounded cells, and both respond similarly to perturbations of the actomyosin cytoskeleton. By contrast, cell–cell junctional complexes and intermediate filaments do not relax tissue stress, but form stable connections between cells, allowing monolayers to behave rheologically as single cells. Taken together, our data show that actomyosin dynamics governs the rheological properties of epithelial monolayers, dissipating applied stresses and enabling changes in monolayer length.
Figure sample
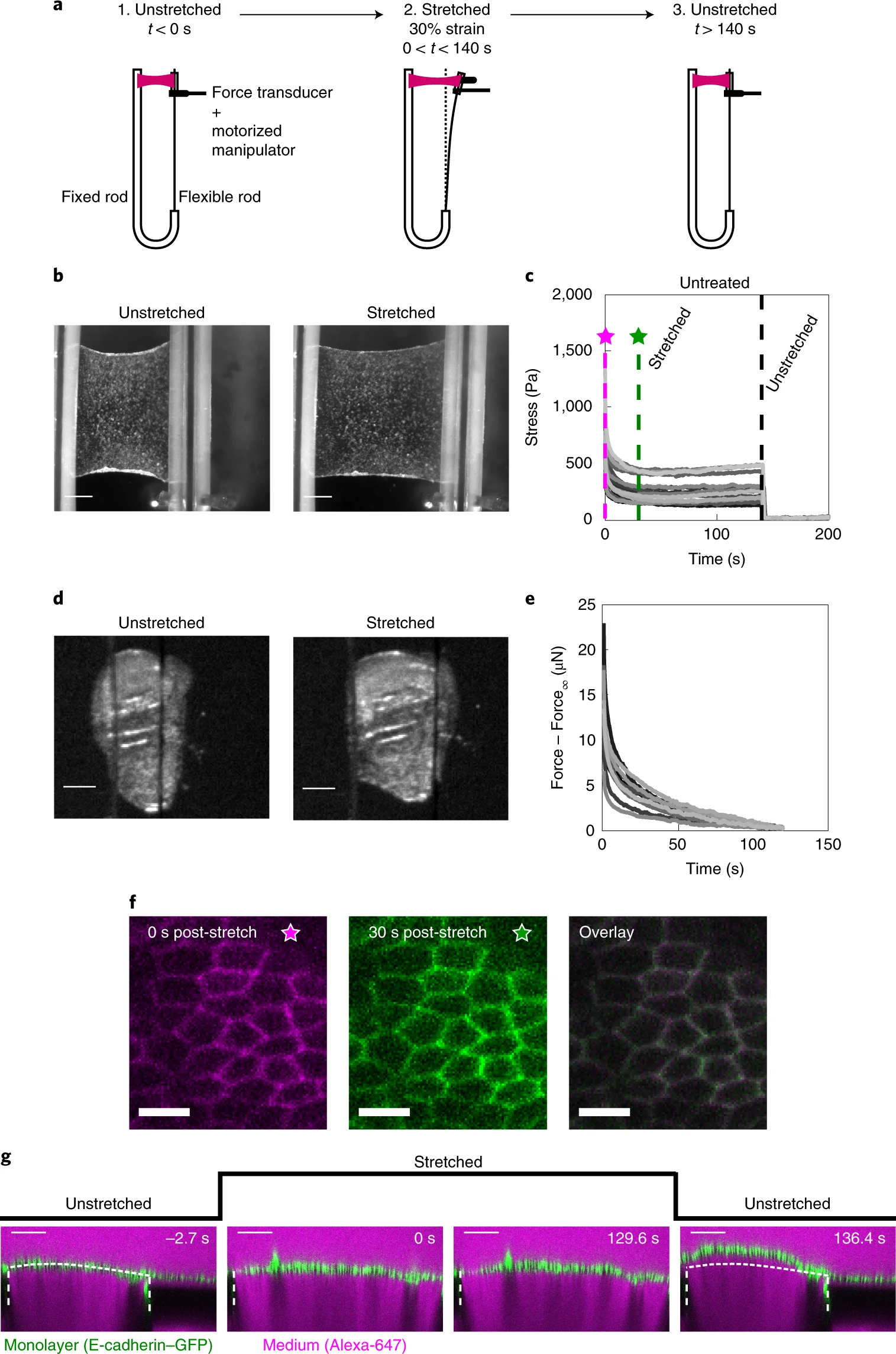
a, Schematic diagram of the stress relaxation experiments. Monolayers were stretched to 30% strain at a 75% s−1 strain rate using a motorized micromanipulator and then kept at a fixed strain for about 130–140 s. The flexible rod was then returned to its initial position and the monolayers were left to recover. b, Bright-field microscopy images of an epithelial monolayer before and during stretch. Scale bars, 0.5 mm. c, Stress relaxation curves of cell monolayers (n = 17). The magenta and green dashed lines indicate 0 s and 30 s after application of stretch. Stresses go to zero on return of the flexible rod to its initial position (t = 140 s, black dashed line). d, Bright-field microscopy images of Drosophila larval wing discs before and during stretch. Scale bars, 100 μm. e, Stress relaxation curves of Drosophila larval wing discs (n = 12). f, Confocal microscopy images of monolayers expressing E-cadherin–GFP (green fluorescent protein) for 0 s (left) and 30 s (middle) after stretch. The two images were overlaid to detect potential cell shape change during relaxation (right). Scale bars, 10 μm. g, Cross-section of a monolayer expressing E-cadherin–GFP before application of stretch (−2.7 s), during stretch (0 s and 129.6 s) and on release (136.4 s). The length of the monolayer on release is different from its length before application of stretch. The glass substrate appears dark due to dye exclusion. The white dashed lines indicate the positions of the glass substrates. The part of the monolayer situated between the two dashed lines is suspended. The dotted white line indicates the shape of the monolayer before application of stretch. Scale bars, 100 μm. This experiment is representative of n = 22 monolayers.


