
Citation
A. Lisica, J. Fouchard, M. Kelkar, T. Wyatt, J. Duque, A.-B. Ndiaye, A. Bonfanti, B. Baum, A.J. Kabla, G.T. Charras
PNAS 119:49 (2022)
Abstract
Abstract
Significance
In growing epithelia, divisions are typically oriented in the epithelial plane to drive tissue expansion. In some organs, divisions are then reoriented to occur perpendicular to the epithelium plane to drive tissue stratification and cell differentiation. When uncontrolled, this switch in orientation can lead to defects in tissue organization and has been suggested to contribute to cancer metastasis. While much is known about the molecular machinery guiding mitotic spindle orientation, less is known about the role of mechanical factors. Here, we show that mechanics plays a role in controlling the plane of division. Overall, our data suggest that the orientation of spindles in the epithelium plane requires a threshold level of mechanical tension at intercellular junctions.
Abstract
The direction in which a cell divides is set by the orientation of its mitotic spindle and is important for determining cell fate, controlling tissue shape, and maintaining tissue architecture. Divisions parallel to the epithelial plane sustain tissue expansion. By contrast, divisions perpendicular to the plane promote tissue stratification and lead to the loss of epithelial cells from the tissue—an event that has been suggested to promote metastasis. Much is known about the molecular machinery involved in orienting the spindle, but less is known about the contribution of mechanical factors, such as tissue tension, in ensuring spindle orientation in the plane of the epithelium. This is important as epithelia are continuously subjected to mechanical stresses. To explore this further, we subjected suspended epithelial monolayers devoid of extracellular matrix to varying levels of tissue tension to study the orientation of cell divisions relative to the tissue plane. This analysis revealed that lowering tissue tension by compressing epithelial monolayers or by inhibiting myosin contractility increased the frequency of out-of-plane divisions. Reciprocally, increasing tissue tension by elevating cell contractility or by tissue stretching restored accurate in-plane cell divisions. Moreover, a characterization of the geometry of cells within these epithelia suggested that spindles can sense tissue tension through its impact on tension at subcellular surfaces, independently of their shape. Overall, these data suggest that accurate spindle orientation in the plane of the epithelium relies on a threshold level of tension at intercellular junctions.
The process of making blood smears is common in both research and clinical settings for investigating the health of blood cells and the presence of blood-borne parasites. It is very often carried out manually. We focus here on smears for malaria diagnosis and research, which are frequently analyzed by optical microscopy and require a high quality. Automating the smear preparation promises to increase throughput and to improve the quality and consistency of the smears. We present here two devices (manual and motorized) designed to aid in the making of blood smears. These are fully documented, open-source hardware, and an important principle was to make them easily fabricated locally anywhere. Designs and assembly instructions are freely available under an open license. We also describe an image analysis pipeline for characterizing the quality of smears and use it to optimize the settings and tunable parameters in the two devices. The devices perform as well as expert human operators while not requiring a trained operator and offering potential advantages in reproducibility and standardization across facilities.
Figure sample
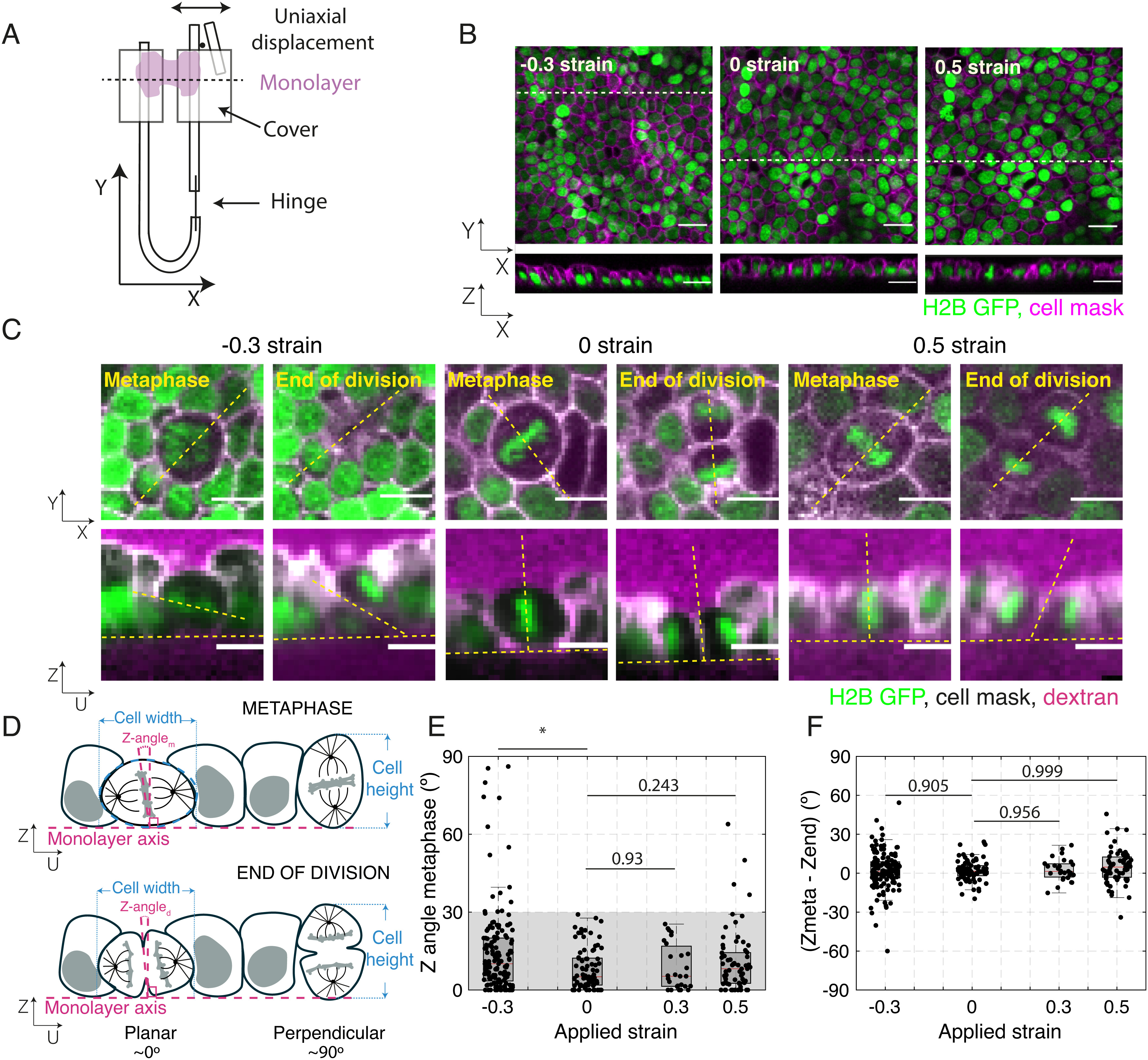
Application of uniaxial compressive strain to epithelial monolayers promotes division out-of-plane. (A) Diagram of the device for mechanical manipulation of suspended MDCK monolayers. The U-shaped device consists of a rigid arm and a flexible arm. Small cover glasses (gray) are glued to the extremities of each arm creating a gap of ~500 μm. A drop of collagen is then polymerized in this gap, and cells are seeded on top of it. Once the cells form a monolayer spanning the gap (magenta), the collagen is removed by enzymatic digestion leaving the monolayer suspended between the two plates. Uniaxial strain can be applied to the monolayer by displacing the flexible arm with a motorized manipulator. (B) Representative images of suspended MDCK cell monolayers subjected to different strains along the X-axis viewed in the XY (Top) and XZ (Bottom) planes. The strain to which the monolayer was subjected is indicated in the Top Left corner. Nuclei are marked with H2B GFP (green) and cell membrane with CellMask (magenta). Dashed white lines indicate the planes at which the XZ profiles were taken. (Scale bar: 10 μm.) (C) Examples of cell divisions in MDCK monolayers subjected to different strains viewed in the XY (Top) and UZ (Bottom) planes. Each cell is shown at metaphase and at the end of cytokinesis. Nuclei are marked with H2B GFP (green), cell membranes are visualized with CellMask (white), and Alexa Fluor 647 dextran (magenta) is added to the medium to allow visualization of the cell outlines. For each cell, the orientation of the spindle defines a UV referential with the U-axis oriented along the pole-to-pole axis (dashed yellow lines) and the V-axis along the metaphase plate (SI Appendix, Fig. S1A). Profile views were taken along the UZ axis. In the profile views, the horizontal yellow dashed lines indicate the plane of the monolayer, while the slanted and vertical dashed lines indicate the orientation of the metaphase plate or the division furrow. (Scale bar: 10 μm.) (D) Diagram of the spindle and division orientation measurements in the UZ plane. The spindle Z-angle at metaphase (Z-anglem) was calculated as the angle between the line passing through the metaphase plate and the line perpendicular to the monolayer plane. Similarly, the spindle Z-angle at the end of division (Z-angled) was calculated as the angle between the line passing through the closed cytokinetic furrow and the line perpendicular to the monolayer plane. (E) Distribution of spindle Z-angles at metaphase (Z-anglem) for different applied strains. Gray shaded area highlights Z-angles <30°. The number of mitotic cells examined for each condition was N = 147 for −30% strain, N = 81 for 0% strain, N = 27 for 30% strain, and N = 68 for 50% strain. Experiments were performed on n = 14 independent days for −30% strain, n = 8 independent days for 0% strain, n = 4 independent days for 30% strain, and n = 8 independent days for 50% strain. The P-value compared with 0% strain was P = 0.003 for −30% strain, P = 0.93 for 30% strain, and P = 0.243 for 50% strain. (F) Difference between spindle Z-angles at the beginning of metaphase and the end of division for each applied strain. The data correspond to the same experiments as in (E). Distributions were compared with 0 with a Z-test. The P value was P = 0.965 for −30% strain, P = 0.992 for 0% strain, P = 0.956 for 30% strain, and P = 0.999 for 50% strain. (E and F) Box plots indicate the 25th and 75th percentiles, the red line indicates the median, and the whiskers extend to the most extreme data points that are not outliers. Individual data points are indicated by black dots.


