
Citation
R. Rezk, B.Z. Jia, A. Wendler, I. Dimov, C. Watts, A.E. Markaki, K. Franze, A.J. Kabla
Neuro-Oncology Advances (2020)
Abstract
Background
Abstract
Background
Glioblastoma (GBM) is a highly aggressive incurable brain tumor. The main cause of mortality in GBM patients is the invasive rim of cells migrating away from the main tumor mass and invading healthy parts of the brain. Although motion is driven by forces, our current understanding of the physical factors involved in glioma infiltration remains limited. This study aims to investigate the adhesion properties within and between patients’ tumors on a cellular level and test whether these properties correlate with cell migration.
Methods
Six tissue samples were taken from spatially separated sections during 5-aminolevulinic acid (5-ALA) fluorescence guided surgery. Navigated biopsy samples were collected from strongly fluorescent tumor cores, a weak fluorescent tumor rim, and non-fluorescent tumor margins. A microfluidics device was built to induce controlled shear forces to detach cells from monolayer cultures. Cells were cultured on low modulus polydimethylsiloxane representative of the stiffness of brain tissue. Cell migration and morphology were then obtained using time lapse microscopy.
Results
GBM cell populations from different tumor fractions of the same patient exhibited different migratory and adhesive behaviors. These differences were associated with sampling location and amount of 5-ALA fluorescence. Cells derived from weak- and non-fluorescent tumor tissue were smaller, adhered less well, and migrated quicker than cells derived from strongly fluorescent tumor mass.
Conclusion
GBM tumors are biomechanically heterogeneous. Selecting multiple populations and broad location sampling are therefore important to consider for drug testing.
Figure sample
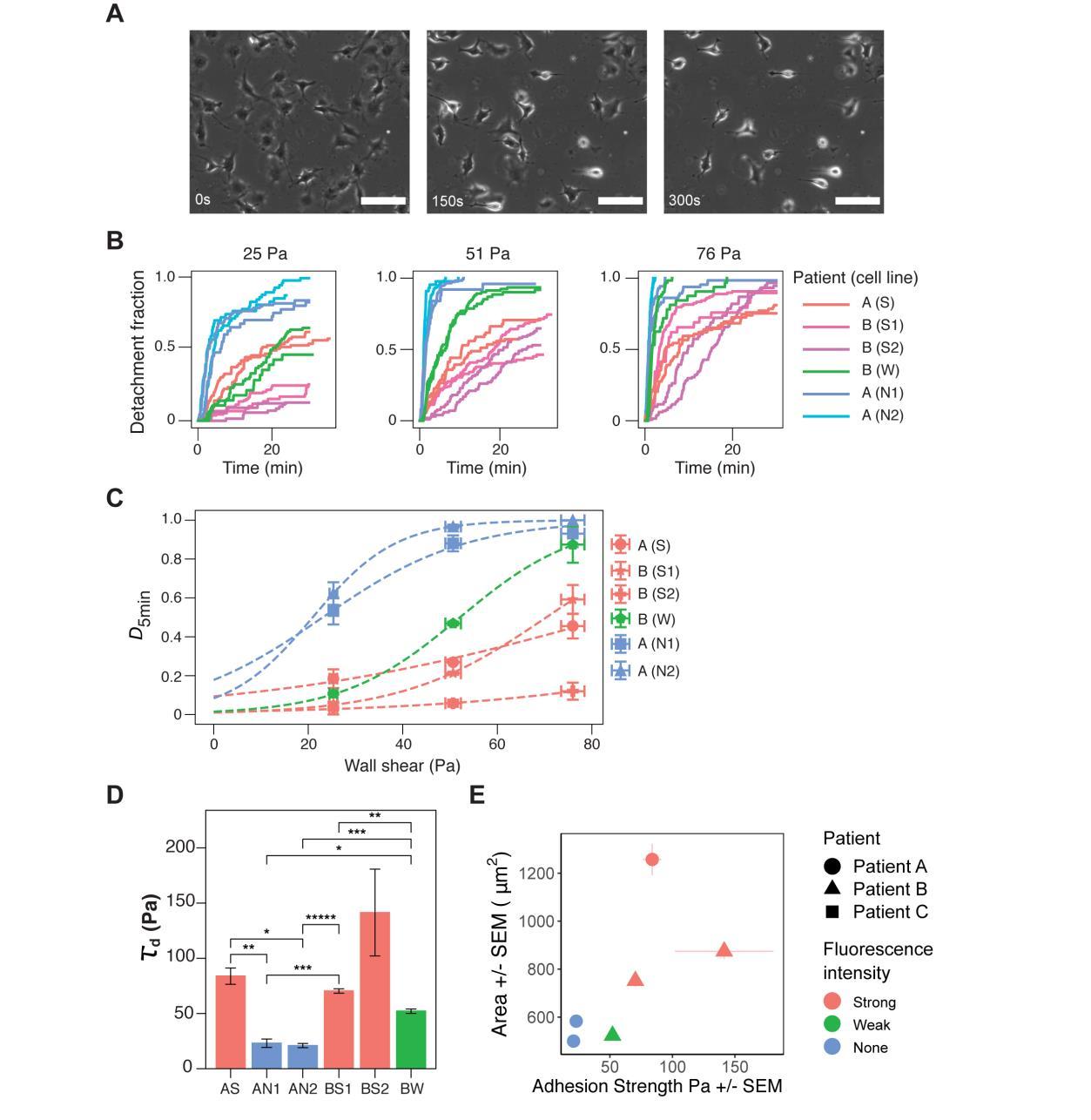
GBM exhibits spatial and inter-tumor heterogeneity in cell-matrix adhesion. (A) Representative phase-contrast images of cells under microfluidic shear detachment at 0, 150, and 300 seconds. Here BW cells are exposed to 0.33 mL/min of flow, corresponding to 51 Pa of shear stress. Scale bars 100 μm. (B) For each condition (shear flow), detachment-time curve were generated using two independent experiments per cell line (25-138 cells per experiment).Detachment increases with shear stress, but the relative ordering of the lines remains constant except for cell line AS. Detachment initially occurs rapidly, and plateaus for most cell lines. (C) The inflection point of each sigmoidal curve was extracted to define τd, a measurement for the cell-matrix adhesion strength of the cell lines. (D) Differences between weak- and strongly-fluorescent lines were statistically significant for all intra-patient pairs except BW and BS2 (one * indicates (*p <0.05) and additional *s indicates further orders of magnitude, under Welch’s t-test with Bonferroni correction). The mean value of τd for patient B was significantly higher than that of patient B (**p = 0.005). (E) Relationship between cell spread area and cell-matrix adhesion strength. Strong fluorescent cells are more adherent and larger than weak- and non-fluorescent cells. Error bars represent SEM from at least two independent experiments.

